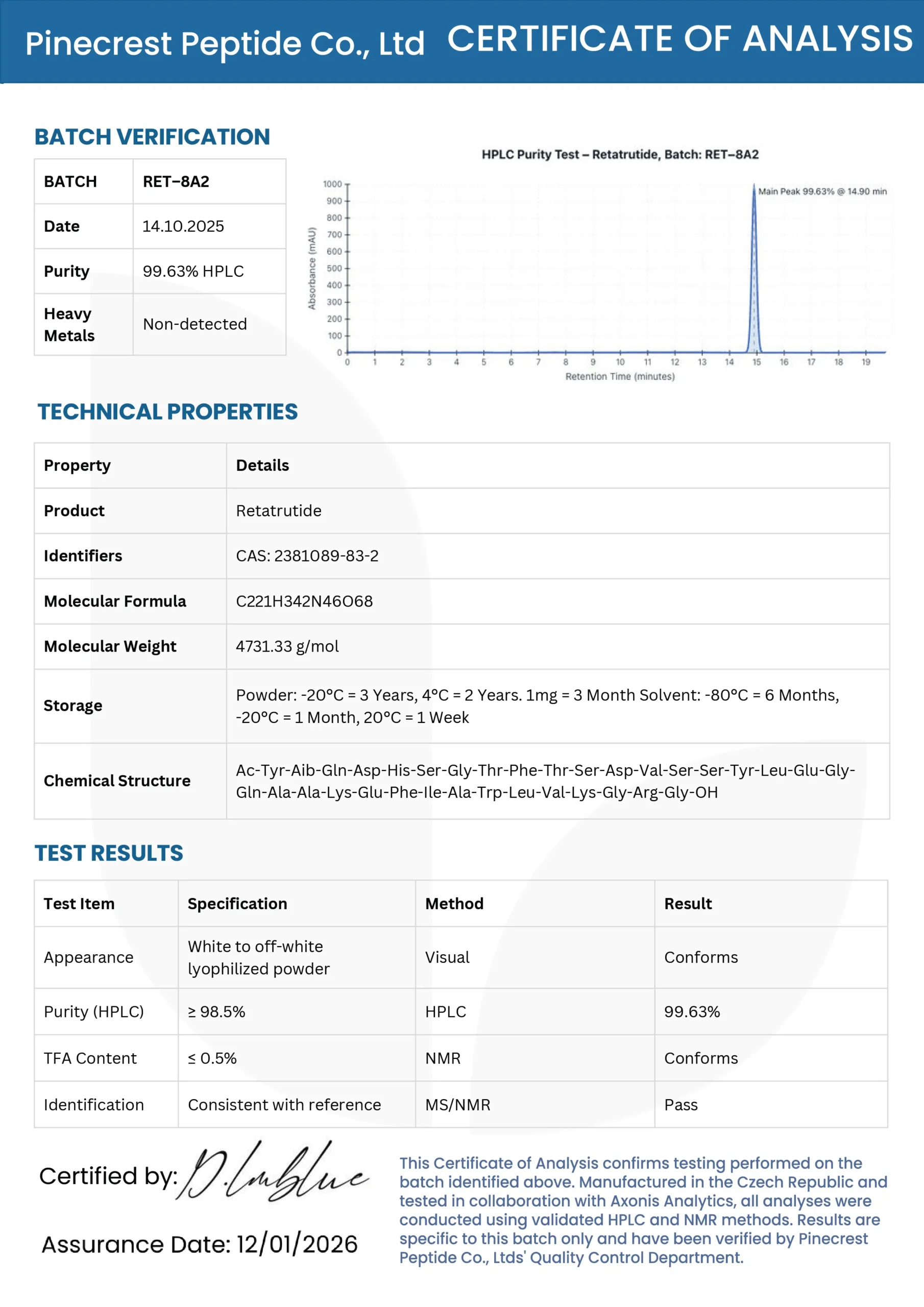
Retatrutide
$85.00 – $310.00Price range: $85.00 through $310.00
Premium Retatrutide for Sale. Retatrutide peptide available in 5mg–60mg vials, 10 vials per box. Premium quality, lab-tested, and shipped safely from the USA.
Premium Retatrutide for Sale – The Next Generation Triple Agonist
The future of metabolic research has arrived.
Retatrutide isn’t just another GLP-1 analog. It’s a revolutionary triple agonist that targets not one, not two, but three distinct receptor pathways simultaneously: GLP-1, GIP, and glucagon . This triple-action mechanism represents the next frontier in metabolic research—and it’s available now from Pinecrest Peptides.
If you’re familiar with semaglutide’s 14.9% weight reduction or tirzepatide’s 17.8% effects in clinical studies, prepare to be impressed . Phase II trials of retatrutide have demonstrated average weight loss of 17.5% at 24 weeks and 24.4% at 48 weeks in overweight and obese adults . That’s a new efficacy ceiling.
When you’re searching for premium Retatrutide for sale, you’re not just buying another peptide. You’re investing in the molecule that’s reshaping how researchers approach metabolic dysfunction, obesity, and related comorbidities. Premium Retatrutide for Sale
At Pinecrest, our Retatrutide:
-
Tests at 99%+ purity (HPLC verified)
-
Delivered as premium lyophilized powder
-
Includes batch-specific Certificate of Analysis
-
Ships from our China facility to your USA door in 5–7 days
-
Backed by our 100% purity guarantee
WHAT IS RETATRUTIDE?
Retatrutide (LY3437943) is a novel synthetic peptide consisting of 39 amino acids, engineered from a GIP peptide backbone to simultaneously activate three distinct receptor types :
-
GLP-1 Receptors (GLP-1R): Reduce appetite, slow gastric emptying, promote glucose-dependent insulin release
-
GIP Receptors (GIPR): Enhance insulin secretion, improve fat metabolism, increase energy expenditure
-
Glucagon Receptors (GCGR): Increase energy expenditure, promote fat oxidation, improve lipid metabolism
This triple agonist approach creates a synergistic interaction among these receptors, resulting in enhanced insulin secretion, improved glucose homeostasis, and refined appetite modulation beyond what single or dual agonists can achieve. Premium Retatrutide for Sale
Structural Excellence – Premium Retatrutide for Sale
What makes retatrutide unique is its sophisticated molecular engineering. The peptide incorporates three non-coded amino acid residues at specific positions :
-
Aib² at position 2: Provides stability against cleavage by dipeptidyl peptidase 4 (DPP4), extending half-life
-
αMeL¹³ at position 13: Promotes glucagon receptor activity
-
Aib²⁰ at position 20: Optimizes GIP activity and pharmacokinetic profile
The result? A molecule with a relatively long half-life of approximately 6 days, supporting once-weekly administration in clinical settings and maintaining steady plasma levels for sustained effectiveness .
Chemical Identity:
-
CAS Number: 2381089-83-2
-
Molecular Formula: C₂₂₁H₃₄₂N₄₆O₆₈
-
Molecular Weight: ~4731.33 g/mol
-
Sequence: Engineered from GIP backbone with strategic amino acid substitutions
MECHANISM OF ACTION: THE POWER OF THREE – Premium Retatrutide for Sale
Triple Receptor Activation
Retatrutide’s magic lies in its ability to engage three complementary pathways :
| Receptor | EC₅₀ Value (Human) | Primary Effects |
|---|---|---|
| GIPR | 0.0643 nM | Insulin secretion, fat metabolism |
| GLP-1R | 0.775 nM | Appetite suppression, glucose regulation |
| GCGR | 5.79 nM | Energy expenditure, fat oxidation |
This balanced activation profile means retatrutide addresses metabolic dysfunction from multiple angles simultaneously :
Appetite Regulation: Through GLP-1 receptor activation, retatrutide influences hypothalamic neurons directly associated with eating behavior, increasing satiety and reducing energy intake .
Energy Expenditure: Glucagon receptor activation leads to significant weight reduction and increased energy expenditure—something GLP-1 alone cannot achieve .
Metabolic Flexibility: GIP receptor activity improves how the body processes and stores nutrients, enhancing overall metabolic health.
Beyond Weight Loss
The triple agonist mechanism shows promise for addressing multiple facets of metabolic syndrome :
-
Glycemic Control: Significant improvements in plasma insulin levels and HOMA-IR
-
Lipid Profiles: Reduction in cholesterol levels
-
Liver Health: Decreased markers of liver damage (AST and ALT), reduced liver hypertrophy
-
Cardiovascular Risk Factors: Potential mitigation through weight reduction and metabolic improvements
-
Fatty Liver Disease: Suppression of genes related to fatty acid synthesis
CLINICAL EVIDENCE: WHAT THE RESEARCH SHOWS
Retatrutide is one of the most extensively studied investigational peptides in recent memory. Here’s what the data shows. Premium Retatrutide for Sale
Phase II Trial Results
A Phase II clinical trial demonstrated remarkable efficacy in overweight and obese adults :
| Time Point | Average Weight Reduction |
|---|---|
| 24 weeks | 17.5% |
| 48 weeks | 24.4% |
To put this in perspective: a 250-pound person losing 24.4% of body weight represents a 61-pound reduction.
Comparative Efficacy in Preclinical Models
A 2026 study published in the International Journal of Obesity compared the efficacy of GLP-1 analogs in MC4R knockout mice—an animal model of genetic obesity . After 21 days of administration:
| Peptide | Body Weight Reduction |
|---|---|
| Semaglutide | 19.7 ± 4.1% |
| Retatrutide | 24.1 ± 5.8% |
| Tirzepatide | 31.6 ± 7.6% |
The study also found that all three GLP-1 analogs improved :
-
Plasma insulin levels
-
HOMA-IR (insulin resistance marker)
-
Cholesterol levels
-
Markers of liver damage (AST and ALT)
-
Liver hypertrophy
Importantly, GLP-1 analogs suppressed genes related to fatty acid synthesis, suggesting potential benefits for non-alcoholic fatty liver disease .
Emerging Research Areas
Obstructive Sleep Apnea and Osteoarthritis: The ongoing TRIUMPH clinical development program (Phase 3) is evaluating retatrutide for the treatment of obesity and two related complications—obstructive sleep apnea (OSA) and knee osteoarthritis (OA) . With over 5,800 participants across four multicenter, randomized, double-blind studies, this program will provide extensive data on retatrutide’s effects beyond weight management .
Chronic Kidney Disease: The TRANSCEND-CKD trial is investigating retatrutide’s effects on kidney function in adults with overweight/obesity and CKD, with and without type 2 diabetes . This Phase 2b mechanistic study will evaluate changes in measured glomerular filtration rate (mGFR) and kidney hemodynamic measurements, potentially opening new therapeutic avenues .
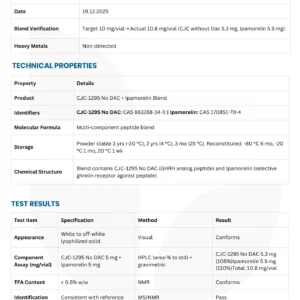
PRODUCT SPECIFICATIONS
| Parameter | Pinecrest Standard | Industry Standard |
|---|---|---|
| Purity (HPLC) | ≥99.0% | ≥98.0% |
| Appearance | White lyophilized powder | White powder |
| Molecular Weight | Confirmed by MS | ~4731.33 |
| Peptide Content | ≥90.0% | ≥80.0% |
| Water Content (KF) | ≤5.0% | ≤8.0% |
| Single Impurity | ≤0.5% | ≤1.0% |
| Total Impurities | ≤1.0% | ≤2.0% |
| Bacterial Endotoxins | ≤10 EU/mg | ≤10 EU/mg |
| pH (1% solution) | 7.0–9.0 | 7.0–9.0 |
| Solubility | ≥5 mg/ml in H₂O | ≥5 mg/ml in H₂O |
| Storage | -20°C (lyophilized) | -20°C |
| Shelf Life | 24 months | 24 months |
Our advantage: We exceed industry standards on purity and moisture content, ensuring your retatrutide remains stable and effective for longer.
ANALYTICAL TESTING: PROVEN BY DATA – Premium Retatrutide for Sale
Every batch of our retatrutide undergoes rigorous quality control testing:
1. High-Performance Liquid Chromatography (HPLC)
-
Quantifies purity with precision
-
Visual chromatogram provided
-
Identifies and quantifies individual impurities
2. Mass Spectrometry (MS)
-
Confirms exact molecular weight
-
Verifies sequence integrity
-
Ensures correct structural identity
3. Karl Fischer Titration
-
Measures moisture content
-
Critical for long-term stability
-
We maintain <5% (industry standard ≤8%)
4. Amino Acid Analysis
-
Confirms correct amino acid composition
-
Validates sequence fidelity
-
Ensures batch-to-batch consistency
5. Endotoxin Testing
-
Ensures ≤10 EU/mg
-
Safe for research applications
-
Verified by validated methods
STABILITY AND HANDLING
Storage Requirements
| Form | Storage Condition | Stability |
|---|---|---|
| Lyophilized powder | -20°C (frozen) | 24 months |
| Lyophilized powder | 2–8°C (refrigerated) | 6–12 months |
| Reconstituted solution | 2–8°C (refrigerated) | ≤30 days |
Protect from light during storage. Retatrutide should not be subjected to repeated freeze-thaw cycles.
Reconstitution Guidelines
For research purposes, retatrutide can be dissolved in sterile water for injection or appropriate buffer. The peptide is soluble at ≥5 mg/ml in pure water . Always use aseptic technique when handling.
RESEARCH APPLICATIONS
Retatrutide is being investigated for multiple research applications :
Primary Research Areas
-
Obesity and Metabolic Research
-
Energy balance studies
-
Appetite regulation mechanisms
-
Body composition analysis
-
-
Type 2 Diabetes Research
-
Glucose homeostasis
-
Insulin secretion pathways
-
Beta-cell function
-
-
Cardiometabolic Research
-
Cardiovascular risk factors
-
Lipid metabolism
-
Inflammatory markers
-
-
Non-Alcoholic Fatty Liver Disease (NAFLD)
-
Hepatic steatosis
-
Liver enzyme modulation
-
Fatty acid synthesis
-
-
Emerging Applications
-
Obstructive sleep apnea (TRIUMPH trials)
-
Knee osteoarthritis (TRIUMPH trials)
-
Chronic kidney disease (TRANSCEND-CKD trial)
-
Typical Research Dosages (For Reference Only)
Note: The following are based on published research protocols and are provided for informational purposes only. All products are for research use only—not for human consumption.
| Research Model | Typical Dosage | Administration |
|---|---|---|
| Murine studies | 0.05 mg/kg | Subcutaneous, daily |
| In vitro assays | Variable | Cell culture |
| Clinical trials | Up to 12 mg | Weekly, subcutaneous |
SAFETY AND REGULATORY STATUS
Current Regulatory Status
Retatrutide is an investigational drug that has not yet received approval from the U.S. Food and Drug Administration (FDA) or other regulatory agencies for therapeutic use . It is currently in Phase III clinical development for obesity, type 2 diabetes mellitus, and non-alcoholic fatty liver disease. Premium Retatrutide for Sale
Research Use Only
Our retatrutide is sold for laboratory research purposes only. It is:
-
Not for human consumption
-
Not for diagnostic or therapeutic use
-
Not FDA-approved for any indication
-
Intended for in vitro studies and analytical applications
Known Adverse Effects from Clinical Trials
Based on published clinical trial data, retatrutide’s side effect profile appears similar to other GLP-1 receptor agonists, primarily gastrointestinal in nature. However, as an investigational drug, its full safety profile continues to be evaluated. Premium Retatrutide for Sale
ORDERING INFORMATION
| Quantity Tier | Catalog | Lead Time |
|---|---|---|
| Sample (1–10 vials) | Inquire | In stock |
| Small Wholesale (10–50 vials) | Inquire | In stock |
| Bulk (50–500 vials) | Inquire | In stock / 3–5 days |
| Enterprise (500+ vials) | Inquire | Negotiated |
Each order includes:
-
Batch-specific Certificate of Analysis
-
HPLC chromatogram (upon request)
-
Mass spectrometry confirmation (upon request)
-
Lot number for full traceability
-
Vacuum-sealed, desiccated packaging
-
Temperature-stable international shipping
Shipping to USA: 5–7 business days (typical)
RETATRUTIDE FAQs
Q: What’s the difference between retatrutide and tirzepatide?
A: Tirzepatide is a dual GIP/GLP-1 receptor agonist. Retatrutide is a triple agonist that adds glucagon receptor activation to the mix . This additional mechanism may contribute to greater energy expenditure and potentially enhanced weight loss effects.
Q: Is retatrutide FDA-approved?
A: Not yet. Retatrutide is currently in Phase III clinical trials for obesity, type 2 diabetes, and related conditions . It has not received FDA approval for any therapeutic use.
Q: What purity should I expect for research-grade retatrutide?
A: Research-grade retatrutide should be ≥98% purity by HPLC, with comprehensive impurity profiling . Pinecrest’s retatrutide is ≥99% with full documentation, including HPLC chromatograms and MS confirmation.
Q: How do I verify the quality of retatrutide?
A: Every batch should come with:
-
Certificate of Analysis with numerical purity
-
HPLC chromatogram (visual purity proof)
-
Mass spectrometry confirmation
-
Moisture content data
-
Endotoxin testing results
We provide all of these with every order.
Q: What’s the molecular weight of retatrutide?
A: The molecular weight is approximately 4731.33 g/mol (C₂₂₁H₃₄₂N₄₆O₆₈) .
Q: How should retatrutide be stored?
A: Lyophilized retatrutide should be stored frozen at -20°C for long-term stability . Refrigerated storage (2–8°C) is acceptable for short-term (weeks). Protect from light.
Q: What’s the half-life of retatrutide?
A: In clinical studies, retatrutide has a relatively long half-life of approximately 6 days, supporting once-weekly administration .
Q: Can retatrutide be used with other peptides in research?
A: In research settings, combinations may be studied, but this requires careful experimental design and appropriate controls. We recommend consulting published literature for validated research protocols.
WHAT RESEARCHERS SAY – Premium Retatrutide for Sale
Dr. Alan Richards – Richards Research Institute, Boston
“Our lab studies metabolic pathways in genetic obesity models. Retatrutide’s triple agonist mechanism offers a unique tool for understanding how different receptor systems interact. Pinecrest’s product has been consistently pure and well-documented across multiple orders.”
Linda Park – Park Research Supply, California
“When retatrutide started gaining traction in the research community, I asked Pinecrest if they could supply it. They already had it in production. Three days later, samples were on the way. That responsiveness is why I’ve stuck with them for years.”
Kevin O’Brien – O’Brien Distributing, Chicago
“My clients are researchers who demand the latest molecules. Retatrutide has been one of our fastest-growing SKUs. Pinecrest’s quality and consistency mean I can stock it with confidence.”
NEED HELP CHOOSING?
Questions about retatrutide for your research? Email us to get Premium Retatrutide for Sale
📧 sales@pinecrestpeptides.com
⏱️ Response time: Within 4 hours
| Retatrutide | 5mg*10vials, 10mg*10vials, 15mg*10vials, 20mg*10vials, 30mg*10vials, 40mg*10vials, 50mg*10vials, 60mg*10vials |
|---|
Be the first to review “Retatrutide” Cancel reply
Related products
Research Peptides
Research Peptides
Research Peptides
Research Peptides for sale
Research Peptides



















Reviews
There are no reviews yet.